Who can break through the billion dollar race track, domestic vaccine giant layout, three RSV products approved in the United States for viruses, drugs, and vaccines
There are new methods to deal with respiratory syncytial virus.
On July 17th local time, the US Food and Drug Administration announced the approval of the monoclonal antibody Nissevir monoclonal antibody for the prevention of RSV related lower respiratory diseases in all infant populations. This vaccine, jointly developed by Sanofi and AstraZeneca, is the first long-term RSV prevention measure that can target all infant populations.
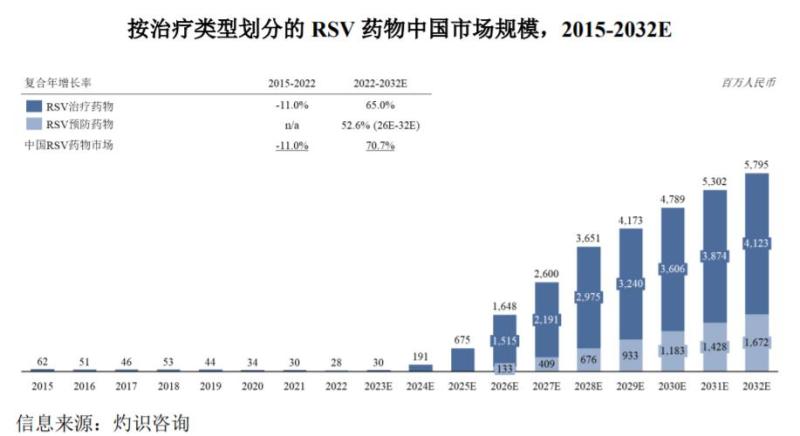
According to data from Zhuo Shi Consulting, the global market size of RSV drugs, including therapeutic and preventive drugs, is expected to increase from $1.9 billion in 2022 to $12.8 billion in 2032, with a compound annual growth rate of 20.8%. In addition to the preventive monoclonal antibody drugs mentioned above, global pharmaceutical giants such as GlaxoSmithKline and Pfizer have successively approved RSV preventive vaccines in the United States since the beginning of this year, and 2023 is also known as the "first year of RSV vaccine commercialization".
From a domestic perspective, the RSV drug market is expected to reach a market size of approximately 4.8 billion US dollars by 2030. At present, no RSV vaccine or drug has been approved in China. From the perspective of research and development layout, the entrants in the relevant field not only include domestic vaccine leading enterprises such as Zhifei Biotechnology and Watson Biotechnology, but also emerging biopharmaceutical companies such as Aike Baifa, Aidi Weixin, and Junshi Biotechnology. Among domestic enterprises, who can first break through the encirclement and win the first RSV disease related product in China is worth looking forward to.
Three products have been approved successively in the United States, and the RSV track abroad is progressing rapidly
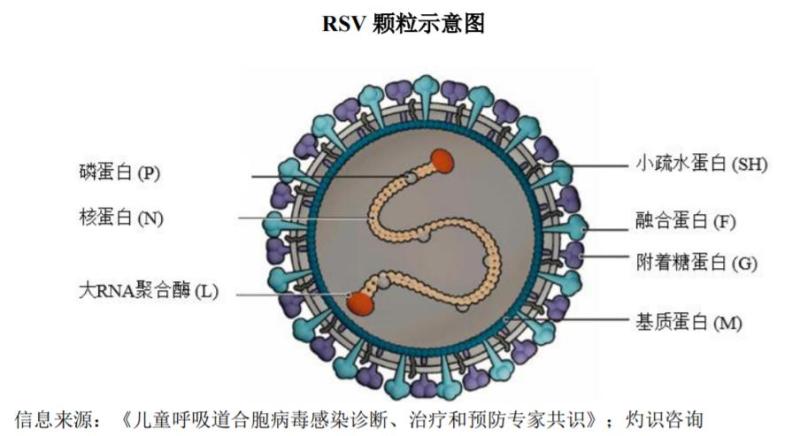
As early as 1956, the RSV virus was isolated from the respiratory tract of chimpanzees. People with normal immunity may self heal after being infected with this virus, but children under 5 years old and elderly people aged 65 and above become susceptible to RSV due to immune dysfunction. Infants may develop lower respiratory tract infections such as bronchiolitis and pneumonia after infection, which is also the primary cause of hospitalization for infants and young children; Elderly infections may lead to serious complications such as chronic obstructive pulmonary disease and congestive heart failure.
For a long time, the clinical treatment options for RSV infected individuals were very limited. Taking children as an example, RSV infection is limited to supportive care and adjuvant treatment, such as oxygen, nasal congestion relievers, nutritional and water supplements, and the use of bronchodilators. The main antiviral treatment methods include ribavirin and interferon, but due to toxic side effects, the use of these two antiviral drugs is more cautious.
In fact, prior to the aforementioned Sanofi/AstraZeneca nixivir monoclonal antibody, the United States had approved the monoclonal antibody drug Palizumab for the prevention of RSV in 1998. However, this drug is only suitable for pediatric patients at high risk of RSV disease, has a small coverage population, and can only reduce hospitalization rates by about 55%, making it expensive.
Nisetumab is suitable for all infant and toddler populations. According to data released by Sanofi/AstraZeneca, the main endpoint of the Phase 2b trial of the drug showed that compared to placebo, Nisetumab significantly reduced the lower respiratory tract infection visit rate of RSV by 70.1%, while demonstrating good safety.
Antibody drugs represented by Nissevir monoclonal antibody can provide protection without activating the immune system. Currently, they are mainly approved for use in infants and young children, while the RSV vaccine that works through the immune system is mainly used in the elderly.
On May 4th this year, GlaxoSmithKline announced that the FDA has approved Arexvy for the prevention of lower respiratory diseases caused by RSV infection in individuals aged 60 and above. This vaccine is the world's first approved RSV vaccine for the elderly, containing a recombinant fusion pre conformational RSVF glycoprotein antigen and GSK proprietary adjuvant AS01.
Pfizer announced on June 6th that the FDA has approved its RSV vaccine ABRYSVO for the prevention of lower respiratory diseases caused by RSV in individuals aged 60 and above. Pfizer emphasizes that ABRYSVO does not contain adjuvants and is composed of two types of preF proteins, which can provide maximum protection against RSVA and B subgroup infections. Its safety and efficacy have been confirmed.
In addition to the two multinational pharmaceutical giants GlaxoSmithKline and Pfizer. Modena's RSV vaccine based on mRNA technology is also expected to be approved soon.
On July 5th local time, Modena announced that its RSV vaccine MRNA-1345 has submitted applications to multiple regulatory agencies worldwide, including starting a rolling submission of the drug's bioproduct license application to the FDA. Earlier, data released by Modner showed that mRNA-1345 achieved the primary endpoint in phase III clinical prevention of RSV infection in the elderly, with a protection rate of 83.7% against two or more RSV-LRTD symptoms and a protective effect of 82.4% against three or more symptoms.
Who is ahead of domestic RSV products such as Zhifei, Watson, and Aike Baifa?
At present, there is no approval for RSV related products in China, and the commercialization field can be described as a blue ocean.
In China, Sanofi/AstraZeneca's Nisetivir monoclonal antibody has been granted breakthrough treatment drug procedures and priority evaluation qualifications by the Drug Evaluation Center of the National Medical Products Administration, and is expected to be approved in China as soon as possible.
In terms of domestic RSV drugs, AK0529, the core product of Aike Baifa, is at the forefront.
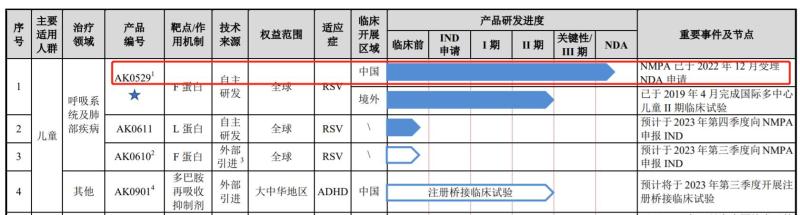
According to the prospectus of Aikebaifa, AK0529 is an oral medication used to treat RSV infections in infants, young children, and adults. The new drug application for the product was accepted by the National Medical Products Administration in December 2022. The successfully completed AirFLO trial of the drug is the world's first phase III clinical trial of an oral RSV antiviral drug to achieve positive results, and the drug is expected to become the world's first approved clinical drug with specific effects against RSV virus infections.
It is worth noting that COVID-19 may have some therapeutic potential for RSV.
On April 17 this year, Junshi Biological announced that the journal Signal Transduction and Targeted Therapy, a subsidiary of Nature, published online the research results of the clinical in vivo efficacy of the oral nucleoside anti COVID-19 drug VV116 as a potential respiratory syncytial virus inhibitor. Research findings. In a mouse model, VV116 has high oral bioavailability, good tissue distribution, and significantly better antiviral effects than ribavirin, and can alleviate pathological damage to lung tissue.
In January this year, the State Food and Drug Administration conditionally approved VV116 for treatment of COVID-19. Junshi Biotechnology stated that the in vivo pharmacological studies published above provide strong evidence for the potential therapeutic effect of VV116 on RSV infection, which will be validated in later clinical studies.
Compared to RSV drugs with preventive or therapeutic properties, the development of RSV vaccines is more difficult. Aike Baifa mentioned in its prospectus that due to the instability of the F protein used by RSV virus as an antigen to invade host cells, the development of RSV vaccines has been difficult and has not made progress for many years. According to information released by relevant domestic enterprises, the domestically produced RSV vaccine is still in its early stages.
On May 23rd, Avixin announced that its research and development team had published Phase I clinical data of the RSV vaccine in the internationally peer-reviewed academic journal Vaccines. ADV110 is an RSV recombinant protein subunit vaccine independently developed by Adivixin, which uses the innovative adjuvant AE011 developed by the company. The vaccine is currently in the phase II clinical trial stage overseas and has completed phase II clinical enrollment in 2022. It is expected to release phase II clinical results in 2023.
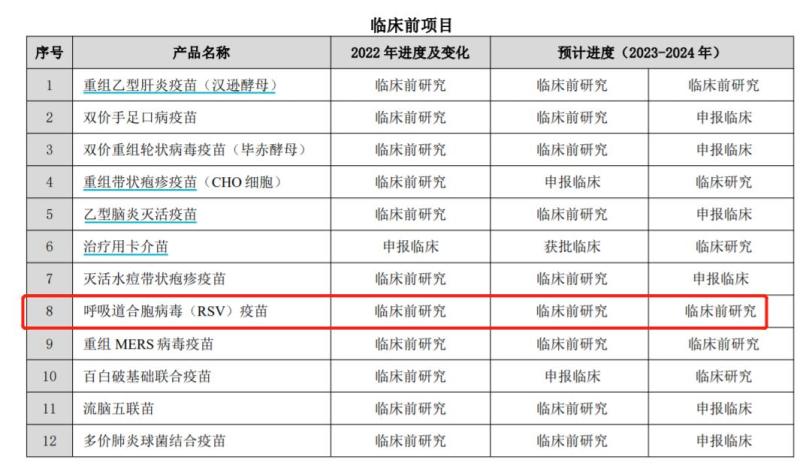
Zhifei Biological's RSV vaccine is currently in a stage where domestic private vaccine giant Zhifei Biological has laid out its presence in the RSV adult vaccine field, but there is not much relevant information. The 2022 annual report shows that the expected progress of the vaccine is in the preclinical research stage. Another leading domestic vaccine company, Watson Biotech, has obtained an RSV vaccine pipeline through cooperation with mRNA technology company. The 2022 annual report of Watson Biology shows that the progressiveness of the company's mRNA vaccine technology platform has been verified in several mRNA vaccines. The company and its partners have jointly developed mRNA RSV vaccines, etc., which are currently in continuous progress. Watson Biotech's partner in the mRNA vaccine field is Shanghai Blue Magpie Biotech.




