Original new drugs generated by artificial intelligence systems entering phase II clinical trials | drugs | new drugs
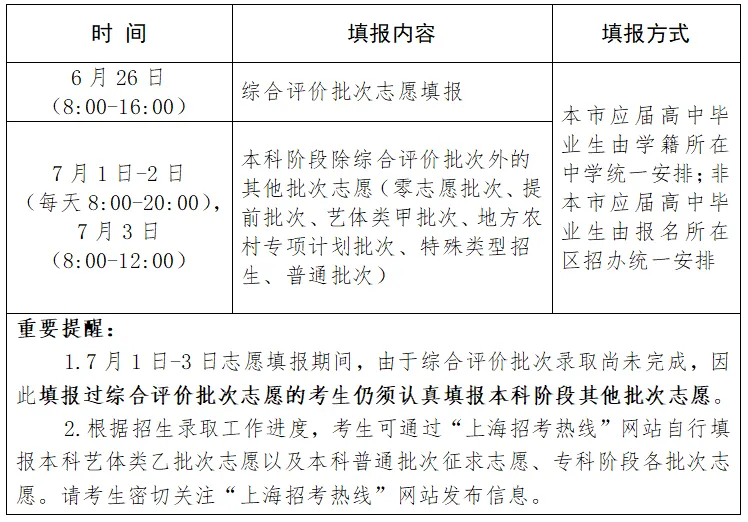
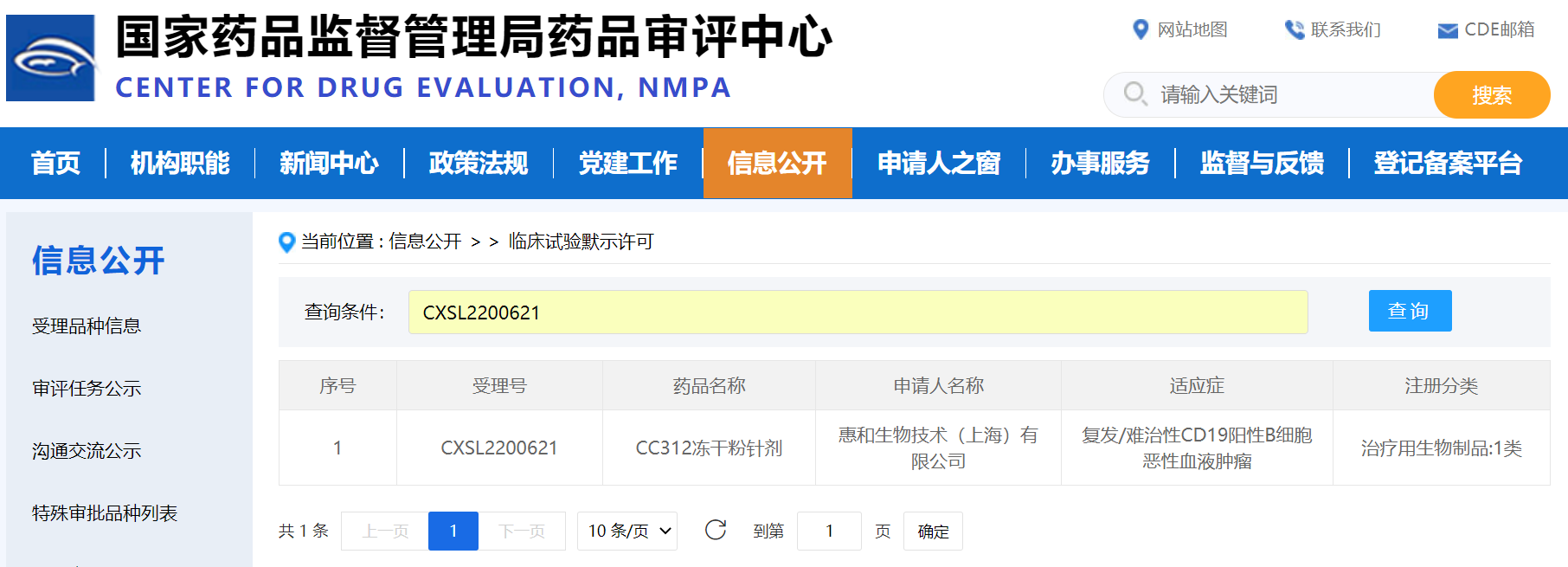
The reporter learned from Yingsi Intelligence yesterday that the candidate drug "INS018:055" developed by a foreign-funded enterprise in Shanghai for the treatment of idiopathic pulmonary fibrosis has completed the first patient administration in phase II clinical trials, marking the first candidate drug to be discovered and designed by generative artificial intelligence for new targets has advanced to the phase II clinical trials.
Professor Xu Zuojun, Chief Physician of Beijing Union Medical College Hospital, Chinese Academy of Medical Sciences, and Lead Researcher of INS018:055 Phase II Clinical Trials in China, introduced that idiopathic pulmonary fibrosis is a chronic scarring lung disease characterized by progressive and irreversible decline in lung function, affecting the lives and health of approximately 5 million people worldwide. Due to the hidden onset and progression of the disease, most patients are diagnosed with a condition that has progressed to the middle and late stages, with a median survival period of 3 years after diagnosis. Given the limited availability of existing therapies and overall poor prognosis, there are still significant unmet clinical needs in the field of IPF.
As an AI driven biopharmaceutical enterprise, Yingsi Intelligence has developed an end-to-end artificial intelligence pharmaceutical system, including target discovery platform PandaOmics, small molecule compound design and generation platform Chemistry42, and clinical trial result prediction platform InClinico.
Yingsi Intelligent "end-to-end" Drug Development Platform Pharma AI
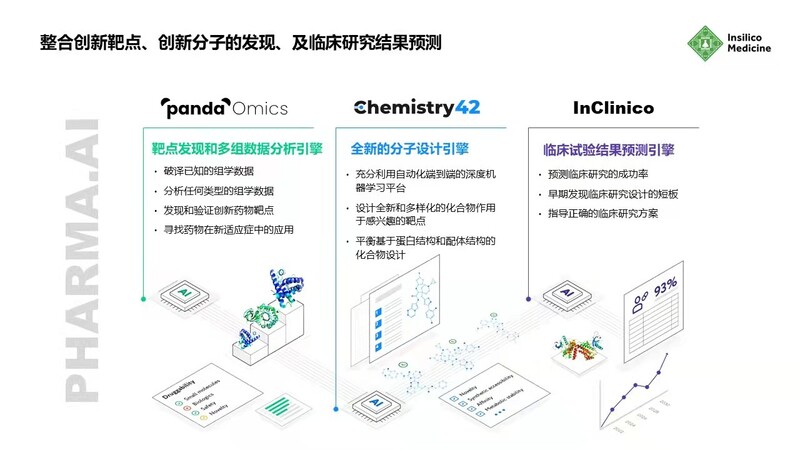
The candidate new drug "INS018:055" for treating idiopathic pulmonary fibrosis was born from this system. Its target is novel and discovered by the PandaOmics platform. This platform analyzes omics data and text data pools to identify genetic differences between patients and healthy populations. Combined with comprehensive analysis of signal pathways, scientific literature, opinion leaders, and other information, it identifies the association between targets and diseases, and then discovers new targets for certain indications.
For the new target, the Chemistry42 platform designed and synthesized a batch of small molecule compounds using deep learning algorithms such as Generative Adversarial Networks. The R&D personnel conducted experimental tests on this batch of compounds and inputted the test data into Chemistry42. After deep learning the data, this platform synthesizes a batch of better compounds. In this way, after several rounds of "design synthesis testing optimization", Yingsi Intelligence identified preclinical candidate compounds.
"INS018:055 has the potential for anti fibrosis and anti-inflammatory effects, and is also the first AI designed drug to enter the clinical stage." Dr. Ren Feng, CEO and Chief Scientific Officer of Yingsi Intelligence, said, "The completion of the first phase II clinical trial for drug administration is an important progress in the AI pharmaceutical field. We look forward to it bringing new choices to patients worldwide."
Dr. Zavorokov collaborated with Dr. Ren Feng to develop a new drug.

It is understood that the Phase II clinical trial used a randomized, double-blind, placebo-controlled method to evaluate the safety, tolerability, pharmacokinetics, and efficacy of "INS018:055" oral administration for 12 weeks in the treatment of idiopathic pulmonary fibrosis in subjects, including four parallel cohorts. In order to test candidate drugs in a larger population, Yingsi Intelligent Plan recruited 60 patients with idiopathic pulmonary fibrosis from nearly 40 research centers in China and the United States, and conducted phase II clinical trials simultaneously.
Dr. Alex Zavorokov, founder and CEO of Yingsi Intelligence, said, "We will explore the effects of drugs discovered and designed by artificial intelligence on patients in clinical trials, which will be a true validation of generative artificial intelligence platforms."