The United States uses the "Sunshine Act" to regulate doctors and pharmaceutical companies too closely, and medical anti-corruption is not a Chinese characteristic bill | Medical | United States
·The particularity of the medical industry means that communication between pharmaceutical companies and healthcare workers is not only a key source of medical innovation, but may also become a public hazard that infringes on public interests and even threatens lives. This also makes regulating the interaction between pharmaceutical enterprises, medical institutions, and healthcare workers the most important ethical issue in the medical community. The Sunshine Act now appears to be the most effective regulatory solution.
With the continuous expansion of domestic medical anti-corruption, the relationship between doctors and pharmaceutical companies such as drugs, medical devices, and consumables has been pushed to the forefront. The marketing costs of domestic pharmaceutical companies ranging from 60% to 80% have attracted widespread attention, and news of pharmaceutical representatives hesitant to enter hospitals has also been circulating from time to time.
In fact, the questioning of the overly close relationship between doctors and pharmaceutical companies is not unique to China and was once a controversial focus in the medical industry in the United States. In order to enhance transparency in the healthcare sector, the Doctor's Compensation Sunshine Act, which was passed in 2010 under the Obama Healthcare Act, stipulates that the economic transactions between pharmaceutical companies and doctors and teaching hospitals must be made public every year.
Since the implementation of the Sunshine Act in 2014, even companies selling only one drug, medical device, or even consumables in the United States are required to report their economic transactions with doctors and hospitals to the government. Nowadays, anyone can search for financial associations between any pharmaceutical company and any doctor on the website of the Federal Medical Insurance and Medicaid Program Service Center in the United States.
The particularity of the medical industry means that communication between pharmaceutical companies and healthcare workers is not only a key source of medical innovation, but may also become a public hazard that infringes on public interests and even threatens lives. This also makes regulating the interaction between pharmaceutical enterprises, medical institutions, and healthcare workers the most important ethical issue in the medical community. The Sunshine Act now appears to be the most effective regulatory solution.
The US Sunshine Act covers pharmaceutical companies whose products are covered by Medicare, Medicaid, and SCHIP government funded insurance programs, including drugs, medical equipment, and consumables. This actually includes almost all pharmaceutical companies in the United States.
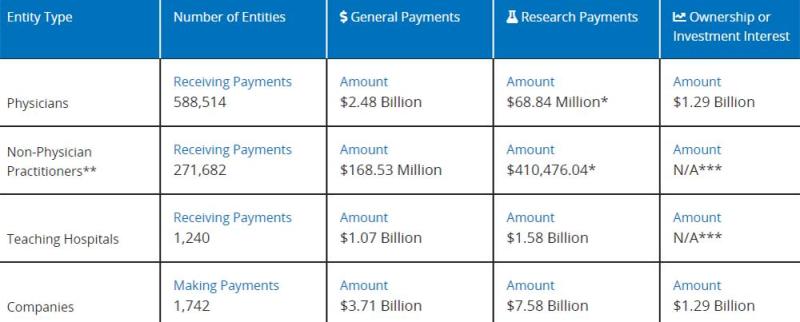
And these pharmaceutical companies need to report any economic transactions with any practicing physician or teaching hospital exceeding $10 to CMS every year. Even if a single transaction is less than $10 and the total amount exceeds $100 per year, it needs to be reported. CMS will organize annual data and publish it. Before the CMS is made public, practicing physicians and hospitals have the opportunity to raise objections and request modifications.
For example, now we can see the latest data up to 2022 on the CMS website:
CMS will organize annual data and publish it. During this year, American pharmaceutical companies paid a total of $2.48 billion in general fees to practicing doctors.
And all of these fees are searchable, which means we can see every payment made by a specific company to a doctor, including the payment date and amount.
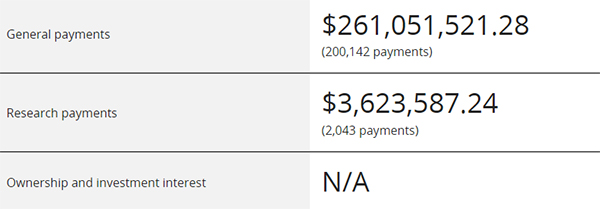
For example, when searching for Pfizer, it will show that in 2022, this pharmaceutical company paid a general fee of $260 million to practicing physicians in the United States, divided into 200000 transactions, with research funding of over $3.6 million for over 2000 transactions.
General fees paid by Pfizer to practicing physicians in the United States in 2022. A simple calculation of the image reveals that the average cost per transaction is only over $100. This is also the situation verified by multiple studies after the implementation of the Sunshine Act: in the United States, the economic exchanges between pharmaceutical companies and doctors are mostly very small.
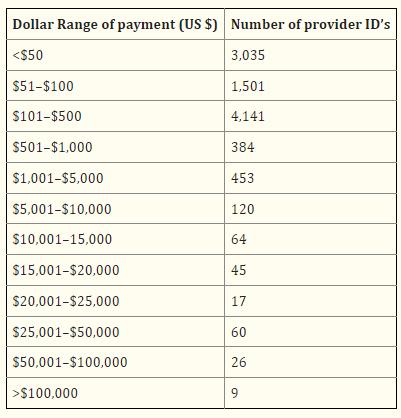
For example, after the first release of data under the Sunshine Act, researchers analyzed the money ophthalmologists received from pharmaceutical companies in 2013 and found that nearly 10000 ophthalmologists had a total of 55996 financial transactions, totaling over 10 million US dollars, with an average single transaction amount of 195.13 US dollars. And the vast majority of doctors receive less than $500 annually from pharmaceutical companies:
In 2013, American ophthalmologists received financial revenue from pharmaceutical companies based on the corresponding number of people in different amount ranges. In 2013, American ophthalmologists spent a total of over 1.6 million dollars on pharmaceutical companies, but this came from nearly 10000 bills, with a maximum of 2500 dollars and an average of 33 dollars. The real bulk comes from consulting fees, exceeding $4.5 million, but the highest single transaction is only $50000, averaging $3313.
These amounts are really nothing compared to the income of American doctors. The original intention of implementing the Sunshine Act in the United States was to make it easy for the public to see if doctors had potential conflicts of interest. However, before the Sunshine Act, many regions, medical associations, and others had detailed regulations on potential conflicts of interest between doctors and pharmaceutical companies, including various public standards. In an environment where healthcare providers have a good overall reputation and no misconduct, the enthusiasm of the general public to check whether doctors have conflicts of interest is also not high.
However, detailed financial transaction records of doctors and pharmaceutical companies facilitate law enforcement agencies to timely monitor criminal activities, including fraud. Sometimes it also brings some unexpected research, such as some people visiting certain specialties to see whether there is gender inequality between male and female doctors in terms of receiving money from pharmaceutical companies.
More importantly, the Sunshine Act fundamentally promotes the standardization of communication between pharmaceutical companies and healthcare workers. Nowadays, Chinese pharmaceutical representatives have been pushed to the forefront, but we cannot ignore the necessity for pharmaceutical companies and medical personnel to operate normally.
These exchanges are not just about pharmaceutical companies promoting products, medicine is constantly developing and innovating, and medical staff need to understand what new drugs and instruments are emerging today, and what changes can be brought to patient treatment. This requires opportunities for communication with pharmaceutical companies. On the other hand, many pharmaceutical innovations also need to seek direction and inspiration from clinical practice.
In China, when it comes to drug prices, it's almost impossible to avoid the popular movie "I'm Not the God of Medicine" from a few years ago. The development of the drug prototype of that movie, Glieves, was not only driven by pharmaceutical companies, but also greatly promoted by American oncologist Brian Druker. Due to poor safety performance in some animal trials, the pharmaceutical company that developed Gliavant was very hesitant to continue making this drug. It was under Druker's coercion that the first clinical trial was launched.
The communication between pharmaceutical companies and doctors like this has positive significance and needs to be encouraged. Under the Sunshine Act, such communication will not be unnoticed.
On the other hand, the situation reflected in China's healthcare anti-corruption efforts. On the one hand, Chinese pharmaceutical companies are innovating in research and development, which requires more communication with doctors. There is not much exchange, but they just rely on their habits to focus on what foreign pharmaceutical companies do. Others do PD-1, follow PD-1, GLP-1, and then GLP-1. So now, when some pharmaceutical research and development media introduce the progress of a new drug internationally, they can see at the end of the article which domestic companies also have layout. On the other hand, unnecessary or unnecessary exchanges such as eating, drinking, playing, and exchanging large amounts of wealth are maintained in various ways.
110 years ago, Louis Brandeis, who later served as a Supreme Court Justice in the United States, once said that sunlight is the best disinfectant. This also applies when regulating the relationship between pharmaceutical companies, medical structures, and medical service providers. To ensure that the relationship between pharmaceutical companies and healthcare workers can maintain long-term health, it is necessary to make this relationship transparent to the public under the sunshine.




