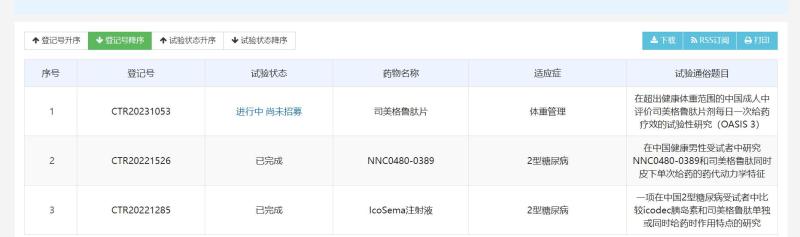
Is Musk's "weight loss miracle drug" about to be approved domestically? The application for the listing of new indications for Smegglutide has been accepted as a divine medicine | injection |? department
Tesla CEO Musk's personally tested "weight loss miracle drug" may further advance its listing pace in China.
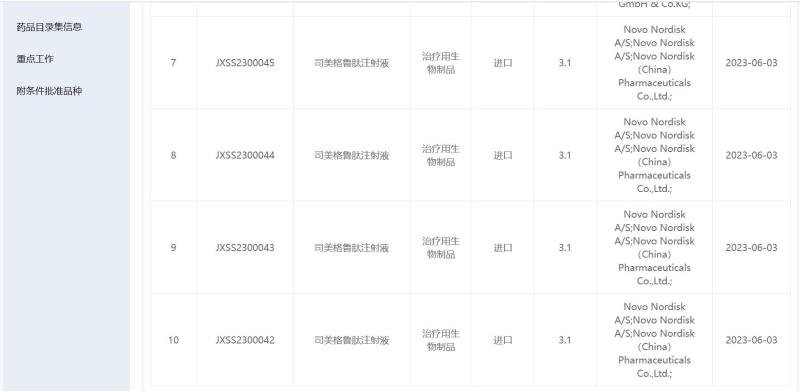
On June 3rd, the official website of the Drug Review Center of the National Medical Products Administration showed that the new indication application for the listing of Smegglutide Injection by the global insulin giant Novo Nordisk has been accepted. The specific content of the indication certificate was not disclosed in the accepted variety information, but the industry generally believes that the indication certificate is likely related to weight loss
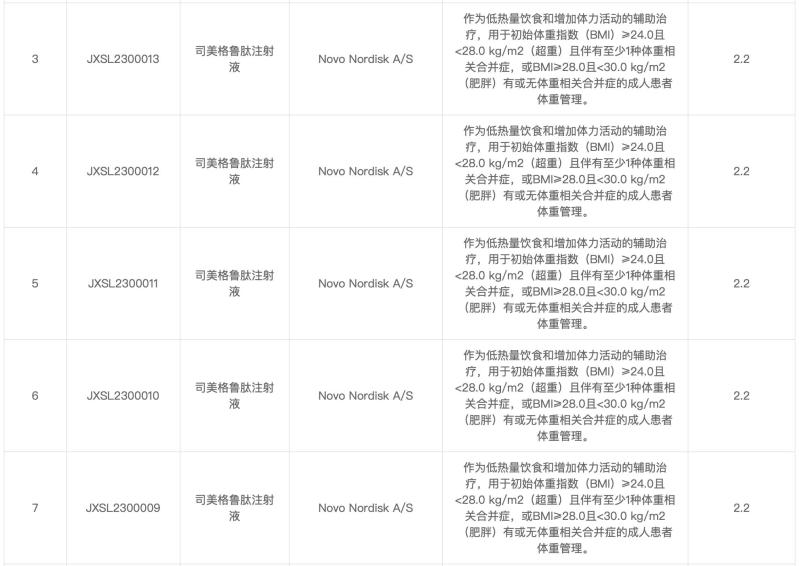
The application for the listing of the new indication for Smegglutide has been accepted. According to the "implied permission for clinical trials," the indication for the previous clinical trial of Smegglutide Injection by Novo Nordisk was: as an adjunctive treatment for a low calorie diet and increased physical activity, for an initial body mass index of ≥ 24.0