Multi pronged tumor ablation, Shanghai's first global medical device approved for market | Technology | Global
Shanghai's medical device innovation has achieved new results. The Shanghai Municipal Medical Products Administration announced today that the registration application for the innovative product of the multimodal tumor treatment system produced by Shanghai Meijie Medical Technology Co., Ltd. has been approved by the National Medical Products Administration. This is the third Class III innovative medical device born in Shanghai this year.
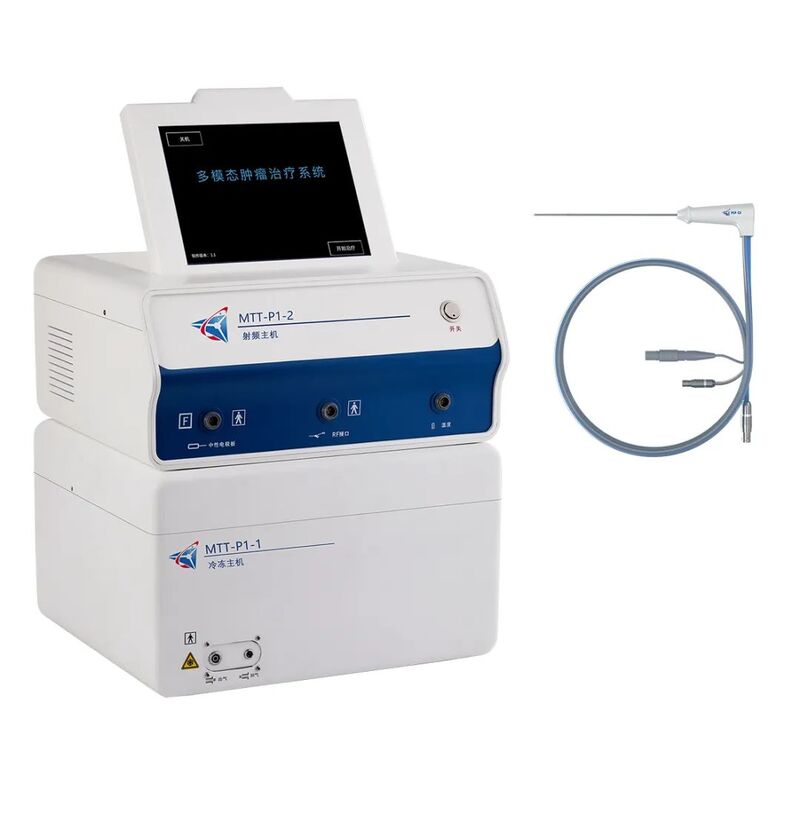
This product is the world's first multimodal tumor radiofrequency therapy system that integrates ultra-low temperature freezing and radiofrequency ablation treatment. It entered the national innovative medical device special review channel in November 2020.
The product pre freezes the target lesion, followed by radiofrequency heating and precise control of the process, to achieve multimodal tumor ablation with overlapping heating and freezing areas, achieving precise treatment of the lesion.
This product achieves precise measurement and real-time feedback of tumor tissue during the treatment process by shielding radio frequency interference during the ablation needle treatment stage; Using high vacuum technology in the non treatment section of the ablation needle, an ultra-thin vacuum insulation layer is constructed to achieve radio frequency electromagnetic field shielding, while completely ablating the tumor and minimizing damage to surrounding tissues. In addition, the integrated control system can achieve visualization of the ablation area by accurately controlling the pre freezing and radio frequency heating processes.
After years of scientific research and exploration, the team led by Xu Xuemin from Shanghai Jiao Tong University has implemented ablation treatment plans through precise control of thermal dose, organic fusion of tissue freezing and radiofrequency heating technology, and for the first time proposed the concept and method of multimodal thermophysical immunotherapy internationally.
In the early stage, Xu Xuemin's team collaborated with Li Wentao's team from Fudan University Affiliated Cancer Hospital to develop a multimodal tumor treatment system that integrates ultra-low temperature freezing and radiofrequency heating, relying on the National Key Research and Development Program of the Ministry of Science and Technology's "Clinical Solution for Image-guided Multimodal Ablation Therapy of Solid Tumors". Through multiple technological iterations and repeated application verifications, the system successfully achieved technological innovation from theory to practice.
The Shanghai Food and Drug Administration has stated that it will strengthen the supervision of products after they are launched to protect the safety of patient equipment.
At present, 81 medical device products in Shanghai have entered the national innovation review channel, and 36 Class III innovative medical devices have been approved for market launch, including the first endoscopic surgical robot, the first domestically produced proton therapy system, and a batch of heavyweight products, all of which have achieved domestic substitution of high-end medical equipment.
In recent years, Shanghai's innovative drugs and innovative medical devices have shown a strong momentum of flying together on both wings. In 2022, a total of 4 Class I domestically produced innovative drugs and 9 Class III innovative medical devices were approved for marketing in Shanghai, ranking first and second in terms of quantity in the country.
The Shanghai drug regulatory authorities, with the help of 12 biopharmaceutical product registration guidance service stations established throughout the city, regularly form groups to visit key characteristic parks, carry out "point by point" training and "face-to-face" Q&A, timely solve problems of enterprises in the research and development, testing, clinical evaluation, registration review and other aspects of medical device products, and strive to solve problems before registration, guiding enterprises to avoid detours.
The Shanghai Food and Drug Administration stated that it will strengthen communication and coordination with the registration and evaluation department of the National Medical Products Administration, actively provide cross disciplinary services with relevant departments, assist in solving the bottlenecks and difficulties encountered by enterprises, stimulate innovation and research and development vitality, shorten the product launch process, and use service-oriented supervision to promote the high-quality development of the Shanghai biopharmaceutical industry.




